In February 2015 the FDA issued a fresh advisory to consumers with peanut allergies: They should avoid foods with cumin, which may be contaminated with undisclosed peanut traces. Less than a week later, an important clinical trial funded by the National Institutes of Health (NIH) revealed that exposing at-risk children to peanut products as infants reduced the chance of developing such allergies later in life by as much as 81%.
Because severe peanut allergies can result in anaphylaxis — a whole-body reaction to an allergen that can be fatal in rare cases — the issue has emerged as a significant public policy issue, prompting legal disputes in certain instances, sometimes dividing parent groups and requiring vigilance on the part of everyone from food plant workers to school cafeteria staff.
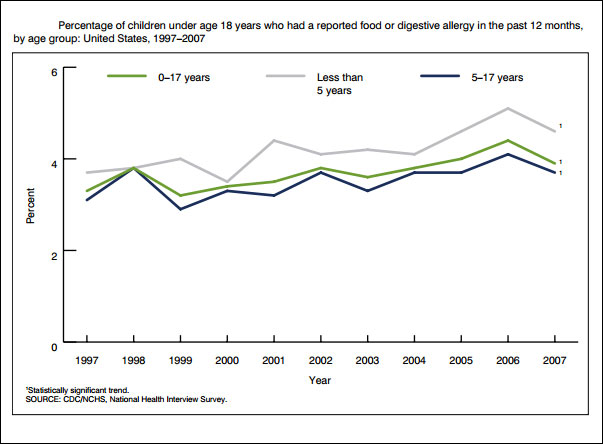
The Centers for Disease Control and Prevention (CDC) notes that the percentage of U.S. children with food allergies of any kind has been growing, and an estimated 4% to 6% now have some kind of food allergy.
A 2010 study published in the Journal of Allergy and Clinical Immunology finds that “peanut allergy, TN [tree nut] allergy, or both continue to be reported by more than 1% of the U.S. population … and appear to be increasingly reported among children over the past decade.” This puts the estimated number of American children with such allergies at more than 3 million. Over the period 1997 to 2008, the prevalence of TN allergy increased from 0.2% to 1.1%, and peanut allergy increased from 0.4% to 1.4%. These rates are also similar to those reported in Canada, the United Kingdom and Australia, the authors note.
The costs of all these trends are not trivial. A 2013 study published in the journal JAMA Pediatrics estimates the annual cost of child food allergies in the United States — including medical costs and lost work and productivity by caregivers — at $24.8 billion, or $4,184 per year per child.
While many have come to accept this growing problem as a fact of life, some still puzzle over how such a common food — a classic staple for generations of peanut butter-and-jelly sandwich-eating kids — has rather rapidly emerged as a potentially dangerous substance requiring this level of public attention. A 2014 paper from researchers at the USDA and UNC Chapel Hill, published in the Annual Review of Food Science and Technology, “Strategies to Mitigate Peanut Allergy: Production, Processing, Utilization, and Immunotherapy Considerations,” reviews the large research literature associated with this phenomenon, providing a solid basis for an understanding of the latest science. The authors — Brittany L. White, Xiaolei Shi, Caitlin M. Burk, Michael Kulis, A. Wesley Burks, Timothy H. Sanders, and Jack P. Davis — examine relevant background and look at potential solutions to this problem.
The paper’s findings include:
- The allergy results when “certain peanut proteins that the immune system mistakenly deems harmful” trigger a set of reactions in two stages, the first of which does not produce an allergic response: “Sensitization” takes place as the digestive system first breaks down the peanuts and plasma cells begin producing immunoglobin E to address what is interpreted as an allergen. In subsequent contact with peanuts, a series of further reactions takes place in the body, which starts releasing inflammatory mediators in the system. Ultimately, this results in “local symptoms such as skin itching/hives, tongue swelling and throat irritation, in addition to various systemic symptoms, such as airway obstruction, blood pressure depression, diarrhea, shock and even fatal anaphylaxis.”
- Peanuts have been cultivated and consumed by humans for centuries, and production in the United States was widespread by the early 1900s. It was not until the 1990s, though, that peanut allergies became an area of focus for researchers and the public, following some well-publicized deaths in the early 1990s.
- The scientific basis for the rise in peanut allergies has not been pinpointed, and this remains a crucial step in alleviating the problem: “Although several interesting hypotheses are being explored, the cause for the rapid emergence of peanut and other food allergies remains unclear. It is likely not one particular factor but a confluence of factors.”
- The medical community has issued conflicting directives as research has advanced: “Dietary avoidance of peanuts and other allergenic foods during pregnancy, breastfeeding and early life was recommended in 2000 (American Academy of Pediatrics, Committee on Nutrition). However, in 2008 this recommendation was withdrawn as ongoing research did not support the idea that early avoidance of an offending food minimized chances of developing a food allergy.”
- Advances have been made in labeling, but there are still areas of ambiguity: “In 2004, the Food Allergen Labeling and Consumer Protection Act (FALCPA) was enacted in the United States to require food manufacturers to declare the source of ingredients derived from the Big-8 allergenic foods in an effort to remove hidden allergens in processed foods…. Additionally, in response to growing consumer concern over shared processing equipment and facilities, the food industry voluntarily includes allergen advisory statements on packaged foods…. Current U.S. legislation, however, applies only to ingredients intentionally added to a food product; it does not define thresholds for food allergens or provide guidance for how the food industry should deal with cross-contact of allergens.”
- Potential solutions are beginning to emerge, but there are still further research and regulatory steps required: “Of the different mitigation strategies being practiced and/or considered for peanut allergy, immunotherapy is of special interest as it targets the source of the problem, adverse immune responses, and therefore minimizes unintended effects on peanut production and utilization. Current immunotherapy trials … are being conducted on relatively small patient sets but are showing excellent promise. Clinical trials are currently underway and larger trials are expected to begin soon. However, additional research studies are needed, and FDA approval will be required before any immunotherapy can be used as treatment.”
The authors conclude that “clear and consistent legislation, policies, practices, etc., are needed for the consumer and producer alike to mitigate food allergy, and these continue to emerge.”
Keywords: nutrition, consumer affairs

Expert Commentary